Compazine
5 mg compazine
Was the problem adequately analysed to include all possible contributory factors from different sectors? These questions should be clearly answered before trying to develop the research proposal medicine cabinets with lights order compazine 5mg with amex. The sections that follow are devoted to giving the guidelines useful for identification, selection, analysis and statement of the given problem. A number of research questions could be presented that may be posed at the various levels of the health system. Discrepancy: All the 500 children at district Y should have completed their vaccination but only 5% out of those who started vaccination have completed. Problem (research) question: why only 5% of the children completed their vaccination? Definite answer: Out of the 1 hospital, 2 health centers and 10 health stations found in district Y only 2 health stations were functioning, the rest were closed due to insecurity in the area. In the above example, assuming that all the given facts are true, there is no need of undertaking a research, since definite answer is obtained to the problem situation. Before deciding on a research topic, each proposed topic must be compared with all other options. The selection and analysis of the problem for research should involve those who are responsible for the health status of the community. This would include managers in the health services, health-care workers, and community leaders, as well as researchers. Relevance: the topic you choose should be a priority problem: Questions to be asked include: How large or widespread is the problem? If the topic has been researched, the results should be reviewed to explore whether major questions that deserve further investigation remain unanswered. Feasibility: Consider the complexity of the problem and the resources you will require to carry out the study. Thought should be given first to personnel, time, equipment and money that are locally available. In situations where the local resources necessary to carry out the project are not sufficient, you might consider sources available at the national level. Political acceptability: It is advisable to research a topic that has the interest and support of the authorities. This will facilitate the smooth conduct of the research and increases the chance that the results of the study will be implemented. Applicability of possible results and recommendations Is it likely that the recommendations from the study will be applied? This will depend not only on the blessing of the authorities but also on the availability of resources for implementing the recommendations. Ethical acceptability We should always consider the possibility that we may inflict harm on others while carrying out research. The above rating should be based on the existing data and not on mere assumptions. In a certain district (population, 150,000), sanitary conditions are very poor (only 5% of households have latrines) and diseases connected with poor sanitation, such as, gastroenteritis and worms are very common. The Ministry of Health has initiated a sanitation project that aims at increasing the number of households with latrines by 20% each year. Discuss about these health problems and rate them based on the selection criteria. When rating these problems based on the criteria, use the rating scale indicated at the bottom of the table (you can also refer to the "Scales for rating research topics presented in section 2. The sections that follow are devoted to giving the principles useful for the analysis and statement of the given problem. Because a clear statement of the problem: Is the foundation for the further development of the research proposal (research objectives, methodology, work plan, etc); Makes it easier to find information and reports of similar studies from which your own study design can benefit; Enables the researcher to systematically point out why the proposed research on the problem should be undertaken and what you hope to achieve with the study results. A list of abbreviations may be annexed to the proposal, but each abbreviation also has to be written out in full when introduced in the text the first time. Describe the reasons for reviewing available literature and other information during the preparation of a research proposal. References that are identified: Should first be skimmed or read Then summaries of the important information in each of the references may be recorded on separate index cards these should then be classified so that the information can easily be retrieved 4. In addition to this, appropriate referencing procedures should always be followed in research proposals as well as in research reports. While reviewing a literature give emphasis to both positive and negative findings and avoid any distortion of information to suit your own study objectives. Finally, after an exhaustive literature review, summarize the findings and write a coherent discussion by indicating the research gap which supports the undertaking of your study. The presentation of research results or scientific publications from other writers without quoting the author is not appropriate. Mention some of the sources of information in your area and describe how such information could be summarized on index cards. For example, if the problem identified is low utilization of health stations in a rural district, the general objective of the study could be to assess the reasons for this low utilization. If we break down this general objective into smaller and logically connected parts, then we get specific objectives. Specific objectives: measurable statements on the specific questions to be answered. Unlike the general objectives, the specific objectives are more specific and are related to the research problem situation. It is said that a question well-stated is a question half-answered, but a question that is poorly stated or unstated is unlikely to be answered at all. Cover the different aspects of the problem and its contributing factors in a coherent way and in a logical sequence. Use action verbs that are specific enough to be measured Examples of action verbs are: to determine to compare to verify 22 Research methodology to calculate to describe to find out to establish Avoid the use of vague non-action verbs such as; to appreciate to understand to study to believe Research objectives can be stated as: A hypothesis is a prediction of a relationship between one or more variables and the problem under study. In order to measure the relationship among variables to be studied the dependent and independent variables need to be identified. The health of children living in rural villagization projects is better than those living in traditional rural communities. To examine whether there is any significant difference between district A and district B with respect to their malaria prevalence rates 23 Research methodology 3. An increase in the frequency of face washing is followed by a reduction in trachoma prevalence One of the most important problems usually observed among students is the tendency of stating too many study objectives which are not appropriately addressed (or sometimes will be forgotten) in the sections that follow. It should be noted that it is on the bases of these specific objectives that the methods, results and discussion sections will be presented. For example, sample size calculations for each stated objective and identifying (selecting) the most appropriate sample size that will answer the required research questions is not covered in the development of most research proposals. It is not uncommon to come across a situation in which some of the specific objectives are not addressed in the results section at all. In most practical situations, the number of specific objectives should not exceed three. Define general objectives, specific objectives and hypotheses by giving your own examples. The objectives of a study should be written after the statement of the research problem and before the methods section. Mention some of the problems that may arise as a result of having too many objectives. Identify the pertinent questions to consider when developing the methodology of a research proposal 2. Describe and understand the various components of the methods section in a research proposal 3. The importance of literature review and formulation of study objectives were also emphasized.
Order compazine once a day
Investigators wishing to focus on test expense may be tempted to report charges rather than costs because charges are more readily available and are generally much higher than costs medicine naproxen purchase compazine on line. However, test charges vary greatly among institutions and may have little relation to what is actually paid for the test or to its actual costs. In many cases, test charges resemble the rack rate on the inside door of a hotel room—a charge much higher than most customers actually pay. On the other hand, estimating how much an institution or society must spend per test is difficult, because many of the expenses, such as laboratory space and equipment, are fixed. One approach is to use the average amount actually paid for the test; another is to multiply charges by the institution’s average cost-to-charge ratio. Intraobserver variability describes the lack of reproducibility in results when the same observer or laboratory performs the test at different times. For example, if a radiologist is shown the same chest radiograph on two occasions, what proportion of the time will he agree with himself on the interpretation? Interobserver variability describes the lack of reproducibility among two or more observers: if another radiologist is shown the same film, how likely is he to agree with the first radiologist? Studies of reproducibility may be done when the level of reproducibility (or lack thereof) is the main research question. In addition, reproducibility is often studied with a goal of quality improvement, either for those making measurements as part of a research study of a different question, or as a part of clinical care. When reproducibility is poor—because either intraor interobserver variability is large—a measurement is unlikely to be useful, and it may need to be either improved or abandoned. Studies of reproducibility do not require a gold standard, so they can be done for tests or diseases where none exists. Of course, both (or all) observers can agree with one another and still be wrong: intraand interobserver reproducibility address precision, not accuracy (Chapter 4). Designs the basic design to assess test reproducibility involves comparing tests done to results from more than one observer or on more than one occasion from a sample of patients or specimens. For tests that involve several steps in many locations, differences in any one of which might affect reproducibility, the investigator will need to decide on the breadth of the study’s focus. For example, measuring interobserver agreement of pathologists about the interpretation of a set of cervical cytology slides in a single hospital may overestimate the overall reproducibility of Pap smears because the variability in how the sample was obtained and how the slide was prepared would not be assessed. The extent to which an investigator needs to isolate the steps that might lead to interobserver disagreement depends partly on the goals of his study. Most studies should estimate the reproducibility of the entire testing process, because this is what determines whether the test is worth using. On the other hand, an investigator who Chapter 12 Designing Studies of Medical Tests 187 is developing or improving a test may want to focus on the specific steps at which variability occurs, to improve the process. In either case, the investigator should lay out the exact process for obtaining the test result in the operations manual (Chapters 4 and 17) and then describe it in the methods section when reporting the study results. The simplest measure of interobserver agreement is the proportion of observations on which the observers agree exactly, sometimes called the concordance rate. However, when there are more than two categories or the observations are not evenly distributed among the categories. For example, if 95% of subjects are normal, two observers who randomly choose which 5% of tests to call ‘‘abnormal’’ will agree that results are ‘‘normal’’ about 90% of the time. A better measure of interobserver agreement, called kappa (Appendix 12A), measures the extent of agreement beyond what would be expected by chance alone. Measures of interobserver variability for continuous variables depend on the design of the study. The best way to describe the data from such a study is to report the mean difference between the paired measurements and the distribution of the differences, perhaps indicating the proportion of time that the difference is clinically important. For example, if a clinically important difference in temperature ◦ is thought to be 0. Other studies examine interobserver or interinstrument variability of a large group of different technicians, laboratories, or machines. These results are commonly summarized using the coefficient of variation, which is the standard deviation of the results on a single specimen divided by their mean, expressed as a percentage. For example, given a coefficient of variation of a serum cholesterol measurement of 2% (2), the standard deviation of multiple measurements with a mean of 200 mg/dL would be about 4 mg/dL and about 95% of laboratories would be expected to report a value between 192 and 208 mg/dL. Confidence intervals for the mean difference should also be avoided because their dependence on sample size makes them potentially misleading. A narrow confidence interval for the mean difference between the two measurements does not imply that they generally closely agree—only that the mean difference between them is being measured precisely. Studies of diagnostic tests can have designs analogous to case–control or cross-sectional studies, whereas studies of prognostic tests usually resemble cohort studies. In the case–control design, those with and without the disease are sampled separately and the test results in the two groups are compared. Unfortunately, it is often hard to reproduce a clinically realistic spectrum of the disease and absence of the disease in the two samples. Those with the disease should not have progressed to severe stages that are relatively easy to diagnose. Those without the target disease should be patients who had symptoms consistent with a particular disease and who turned out not to have it. Studies of tests that sample those with and without the target disease separately are also subject to a bias in the measurement of the test result if that measurement is made knowing whether the sample came from a case or control. Finally, studies with this sampling scheme cannot be used (without other information) to estimate predictive value or posterior probability (discussed below). Therefore, ‘‘case–control’’ sampling for diagnostic tests should be reserved for rare diseases for which no other sampling scheme is feasible. Asinglecross-sectional sample of patients being evaluated for a particular diagnosis generally will yield more valid and interpretable results. Because the subjects were enrolled before it was known whether they were bacteremic, the spectrum of patients in this study should be reasonably representative of patients who present to emergency rooms with fever. A variant of the cross-sectional sampling scheme that we call tandem testing is sometimes used to compare two (presumably imperfect) tests with one another. Both tests are done on a representative sample of patients who may or may not have the disease and the gold standard is selectively applied to the patients with positive results on either or both tests. Because subjects with negative results may be false-negatives, the gold standard should also be applied to a random sample of patients with concordant negative results. This design, which allows the investigator to determine which test is more accurate without the expense of measuring a gold standard in all the subjects with negative test results, has been used in studies comparing different cervical cytology methods (4). Prognostic test studies require either prospective or retrospective cohort designs. In prospective cohort studies, the test is done at baseline, and the subjects are then followed to see who develops the outcome of interest. Then the viral load can be measured in the stored blood, to see whether it predicts prognosis. The nested case–control design (Chapter 7) is particularly attractive if the outcome of interest is rare and the test is expensive. Although it is simplest to think of the results of a diagnostic test as being either positive or negative, many tests have categorical, Chapter 12 Designing Studies of Medical Tests 189 ordinal or continuous results. Whenever possible, investigators should use ordinal or continuous results to take advantage of all available information in the test. Most tests are more indicative of a disease if they are very abnormal than if they are slightly abnormal, and most also have a borderline range in which they do not provide much information. The outcome variable in a diagnostic test study is often the presence or absence of the disease, best determined with a gold standard. Wherever possible, the assessment of outcome should not be influenced by the results of the diagnostic test being studied. This is best accomplished by blinding those measuring the gold standard so that they do not know the results of the test. Sometimes uniform application of the gold standard is not ethical or feasible for studies of diagnostic tests, particularly screening tests. Women with positive mammograms were referred for further tests, eventually with pathologic evaluation as the gold standard. However, it is not reasonable to do biopsies in women whose mammograms are negative.

Order compazine with mastercard
Under a parallel design medications diabetes order online compazine, treatment comparison is made based on both inter-subject and intra-subject variabilities, whereas treatment comparison is made based on the intra-subject variability under a crossover design under appropriate statistical assumption. As it can be seen, the sample size could be reduced by 30% by switching a parallel design to a crossover design when ρ =0. Any slight or moderate deviations from these initial values could result in a substantial change in the calculated sample sizes. Thus, it is suggested that a sensitivity analysis with respect to these initial values be performed. Sensitivity analysis provides useful information regarding what to expect if a deviation in any of the initial values shall occur. Peopleinpracticemaywanttoseehowmuchthesamplesizewould increase when the variability increases, which is equivalent to study how much sample size would be saved if the variability decreases. For evaluation of treatment effect based on discrete clinical endpoint, the proportions of events that have occurred between treatment groups are often compared. Under a given study design, statistical tests for specific hypotheses such as equality or equivalence/non-inferiority can be carried out based on the large sample theory in a similar manner as continuous responses discussed in Chapter 3. In this chapter, our primary focus will be placed on comparingproportions between treatment groups with binary responses. In the next section, a general procedure of power analysis for sample size calculation for testingone-sample problem is given. Formulas for sample size calculation for comparingrelative risks between treatment groups under a parallel design and a crossover design are given in Section 4. In clinical research, xi could be the indicator for the response of tumor in cancer trials, i. Without loss of generality, consider >0(<0) an indication of improvement (worsening)ofthe test drugas compared to the reference value. In what follows, formulas for sample size calculation for testingequality, non-inferiority/superiority, and equivalence are derived. The formulas provide required sample sizes for achievinga desired power under the alternative hypothesis. One-Sample Design 85 Under the alternative hypothesis that p = p0 +,where =0,thepower of the above test is approximately √ n| | Φ − zα/2. For example, if we know that p ≤ p˜,1− p ≤ p˜,and 2 ≥ ˜2,where˜p is a known value between0and0. When p − p0 = δ, the test statistic √ n(ˆ− δ) pˆ(1 − pˆ) approximately has a standard normal distribution for large n. Thus,we reject the null hypothesis at the α level of significance if √ n(ˆ− δ) >zα. Large Sample Tests for Proportions If >δ, the power of the above test is given by √ n(− δ) Φ − zα. The proportion of the responses is concluded to be equivalent to the reference value of p0 if the null hypothesis is rejected at a given significance level. The above hypotheses can be tested usingtwo one-sided test procedures as described in Chapter 3. The null hypothesis is rejected at approximately α level of significance if √ √ n(ˆ− δ) n(ˆ+ δ) < −zα and >zα. Suppose in addition to the study of the change in bone density post-treatment, it is also of interest to evaluate the treatment effect in terms of the response rate at the end of the study. Sample size calculation can then be carried out based on the response rate for achievinga desired power. The definition of a responder, however, should be given in the study protocol prospectively. Test for Equality Suppose that the response rate of the patient population under study after treatment is expected to be around 50%. Also assume that a difference of 10% in responder rate is considered of no clinical significance (δ = −10%). Large Sample Tests for Proportions Test for Equivalence Assume that one brand name drugfor osteoporosis on the market has a responder rate of 60%. It is believed that a 20% difference in responder rate is of no clinical significance. Hence, the investigator wants to show the study drug is equivalent to the market drug in terms of responder rate. Then, which one should be used is always a dilemma because one is not necessarily more powerful than the other. The same idea can be applied to the testingproblems of non-inferiority/superiority. In practice, pi is usually estimated by the observed proportion in the ith treatment group: ni 1 pˆi = xij. Without loss of generality, consider >0(<0) an indication of improvement (worsening) of the test drugas compared to the control value. In what follows, formulas for sample size calculation to achieve a desired power under the alternative hypothesis are derived for testingequality, non-inferiority/superiority, and equivalence. When δ>0, the rejection of the null hypothesis indicates the superiority of the test drug over the control. As it is well known, gram-positive and gram-negative pathogens are commonly associated with skin and skin structure infections such as streptococci, staphylococci, and various strains of enterobacteriaceae. For the evaluation of the effectiveness of a test antibiotic agent, clinical assessments and cultures are usually done at a post-treatment visits. If the culture is positive, the pathogen(s) is usually identified and susceptibility testingis performed. The effectiveness of therapy is usually assessed based on clinical and bacteriological responses at post-treatment visit. Large Sample Tests for Proportions the original site of infection in the absence of local or systemic signs of infection at post-treatment visits), and failure. Suppose that a pharmaceutical company is interested in conductinga clinical trial to compare the efficacy, safety, and tolerability of two antimicrobial agents when administered orally once daily in the treatment of patients with skin and skin structure infections. In what follows, we will consider the situations where the intended trial is for (i) testingequality of mean cure rates, (ii) testingnon-inferiority or superiority of the test drug as compared to the active control agent, and (iii) testing for therapeutic equivalence. Test for Equality In this example, suppose that a difference of = 20% in clinical response of cure is considered of clinically meaningful difference between the two anti-microbial agents. Test for Non-Inferiority Now, suppose it is of interest to establish non-inferiority of the test drugas compared to the active control agent. Also, suppose the true mean cure rates of the treatment agents and the active control are 85% and 65%, respectively. Two-Sample Parallel Design 93 Test for Superiority On the other hand, the pharmaceutical company may want to show superiority of the test drugover the active control agent. As it can be seen, testingsuperiority usually requires larger sample size than testingnon-inferiority and equalty. Test for Equivalence For establishment of equivalence, suppose the true cure rate for the two agents are 75% (p1 =0. Which test (conditional/unconditional) should be used is always a problem because one is not necessarily always more powerful than the other. However, a drawback of the conditional approach is that it is difficult to be generalized to other testing problems. As a result, in this section and also the followingsection, we will focus on the unconditional method because it provides a unified approach for all the testingproblems mentioned above. Nevertheless, for the purpose of completeness, the conditional approach for a two-sample test of equality is also presented below. As a result, the sample size needed for achievinga power of 1−β can be obtained by solvingthe followingequation | | (p1(1 − p1)/n1 + p2(1 − p2)/n2) (1/n1 +1/n2)p(1 − p) −zα/2 = zβ. Two-Sample Crossover Design 95 this leads to n1 = κn2 n = 1 [z (1 + 1/κ)p(1 − p)+z p (1 − p)/κ + p (1 − p)]2. Note that the observations from the same subject can be correlated with each other. By specifyingthat P(xijkl =1)=pk, it implies that there are no sequence, period, and crossover effects. The statistical model incorporates those effects are more complicated for binary data compared with continuous data. Let = p2(test) − p1(reference), 1 x¯ijk· = (xijk1 + ···+ xijkm and dij =¯xij1· − x¯ij2·.

Discount compazine 5mg on-line
Egan’s first article emphasized how his protocol could help decide medications causing thrombocytopenia buy compazine with a visa, for instance, whether a lump discovered through a physical exam warranted a surgical biopsy. His second article did suggest mammographic screening of patients who had been successfully treated (to check for recurrence of the disease) but did not propose screening the general population. Egan went on to participate in research that showed his techniques could be used by other radiologists. In 1965, after moving from Anderson to Emory University in Atlanta, Georgia, he continued to enthusiastically promote mammography, lecturing widely and training radiologists and technicians well into the 1970s. However, Egan focused his research mainly on testing women who had breast complaints or prior histories of breast cancer rather than on population-wide screening (although, as we will see, he did help adapt his protocol for rapid, high-volume screening). Anderson Hospital and Tumor Instituteb in Houston, Texas, developed Leborgne’s technique in a different direction. In 1956, Anderson’s chief radiologist had asked Egan, then a new resident at the hospital, to investigate X-ray diagnosis of breast cancer. In about five years, Egan developed and tested protocols that specified: 1) lower power X-rays than those used for other kinds of diagnostic applications. This protocol, described in papers published in 1960 and 1962, had a ninety-seven percent correspondence with results produced by traditional diagnostic procedures. As Egan started developing his protocols, another radiologist, Jacob Gershon-Cohen, was X-raying the breasts of women with breast cancers at Albert Einstein Medical Center in Philadelphia. Gershon-Cohen intended to record changes in breast tissues that occur as the disease advances. However, the Philadelphia radiologist became interested in early detection and, in the late 1950s, X-rayed the breasts of over 1,300 women who displayed no symptoms. In 1961, he published results that, like Egan’s research suggested, Xrays could detect malignant tumors at a very early stage. The results of mammography performed by the radiologists after they returned to their own hospitals also had a high correspondence with traditional diagnoses – seventy-eight percent. Screening, which included physical examinations and patient histories as well as mammograms, was to be repeated annually for four years. They also used a simpler, faster, mammographic procedure designed by pioneer Robert Egan. Egan also helped train the radiologists and technicians who performed the screening in the trial. Only thirty-one women who had been screened had died of breast cancer -forty percent less than the fifty-two women who had died of breast cancer in the control group. They entail taking a sample of cells on a swab from the opening at the bottom of the uterus and testing the cells in a lab for signs of cancer. Pap smears were the first large-scale attempt at early detection, and they provided a model for large-scale screening of asymptomatic women for breast cancer. See Table 1) And, because treatment after early detection did not cure all breast cancers, Bailar estimated mammography alone prevented only about a dozen deaths (out of the forty-four additional early detections). Meanwhile, mammography could itself have induced sixteen cancer deaths by exposing more than 20,000 healthy women to radiation (according to Bailar’s estimates). Note: Bailar did not consider the slightly higher number of cancers reported in the screened group to be significant. By 1977, the researchers reported a variety of problems, including mistaken diagnoses that had led to fifty-three unnecessary mastectomies. The act increased funding for research and development of cancer diagnostics and treatments. The initiatives supported by this act are often referred to as part of then-President Richard Nixon’s War on Cancer. However, attendees at the conference could not agree on how often women should be screened and whether women should begin screening before menopause (which typically starts around age fifty). Although the equipment had been designed to reduce the training necessary to produce sharp Xrays, it also happened to reduce radiation exposure. Possibly because mammography was not yet a common procedure, no other companies were selling specialized equipment when free screening was started in the U. However, the controversy about screening that erupted in 1976 discouraged manufacturers from marketing or improving specialized equipment. As mentioned, Egan’s protocols had specified specialized X-ray film and low-powered X-rays. The specifications -like Gros’s specialized equipment -happened to reduce radiation exposure but were not always followed: some radiologists and technicians used standard X-ray film and power settings that reduced accuracy and exposed patients to more radiation. Instead they used plates intended for general X-ray use and popularized in 1971 by the copier pioneer Xerox. The plates, which Xerox had developed as a substitute for traditional X-ray film, enabled X-ray images to be printed on paper. And, like standard film, they exposed patients undergoing mammography to more radiation than specialized film. Unlike Robert Egan, who not only developed his mammography protocol but also then devoted himself to promoting its widespread use, Charles Gros had largely focused on developing equipment. By the mid-1970s, only Sweden and Scotland had trials for screening mammography under way. However, besides Austria, other European countries did not start screening programs. Xonics’ devices adapted standard X-ray equipment and were intended to produce chest X-rays and mammograms. Lacking its own sales and marketing, Xonics acquired Standard X-ray, a longtime American X-ray manufacturer, in 1975. And, despite promising results in studies of its mammography units, Xonics failed to secure more than a few hundred orders. In 1983, the company sold out to Elscint, an Israeli medical imaging company, and by 1985, Elscint had shut down the business it had acquired from Xonics. A small-scale breast cancer screening program launched in 1977 relied on physical exams. Radiologists did sometimes X-ray breasts after discovering abnormalities in physical exams. However, Japanese physicians usually preferred ultrasoundh technology -developed by Japanese researchers and Japanese companies -when diagnosing breast cancer. Research later showed that dense breast tissues reduced the accuracy of mammograms. And, although dense breast tissue is more common in Japan, it is also found in women in other parts of the world. Except for one startup (See box Xonics’ Mammography System ), all the other companies that started selling mammography equipment in the U. When Hitachi developed breast imaging equipment, it invested primarily in ultrasound technology rather than X-ray. However, these images did not provide much information about the precise location of lumps. Therefore, to perform a biopsy – the next step in diagnosing potential tumors -surgeons had to cut open breasts extensively. Previously, patients sat upright making it difficult to remain still during diagnostic procedures. Alternatively, radiologists could themselves extract suspect cells using a very fine needle instead of having surgeons cut open breasts. However, although the fine needle procedures were minimally invasive and produced less discomfort and scarring, they required more expertise, which many radiologists did not have. Therefore, the Swedish innovation did not significantly change normal diagnostic practice in the 1970s – and besides Tekniska Rontgencentralen, no other producer offered stereoscopic imaging equipment at that time. Thus, diagnostic equipment and screening equipment (that did not produce stereoscopic images and continued to position patients upright) would emerge as distinct categories. Use Broadens (1980s) New Equipment and Rules In the 1980s, companies developed equipment with controls and filters that (when used with newlyavailable, more sensitive film) reduced radiation exposure. The improvements also reduced the risks of misdiagnosis by producing more detailed images. In addition, companies introduced basic, low-end devices priced at $16,000 in 1989, down from $65,000 in the early 1980s. These standards and inspections forced hospitals to stop adapting general purpose X-ray equipment for mammography, as many had previously done.

Buy cheap compazine
Disagreements commonly arise among the research team regarding who should be an author or the order of authors medicine overdose buy cheap compazine 5 mg on line. These issues are best discussed explicitly and decided at the beginning of a project. Collaborators subsequently might not carry out the tasks they agreed to , for example, failing to carry out data analyses or prepare 234 Implementation a first draft. Changes in authorship should be negotiated when decisions are made to shift responsibilities for the work. Detailed suggestions have been made on how to carry out such negotiations diplomatically (32). Conflicts of Interest Researchers may have conflicting interests that might impair their objectivity and undermine public trust in research (33,34). An investigator may be the personal physician of an eligible research participant. Such participants might fear that their future care will be jeopardized if they decline to participate in the research, or they may not distinguish between research and treatment. Furthermore, what is best for a particular participant may differ from what is best for the research project. In this situation, the welfare of the participant should be paramount, and the physician must do what is best for the participant. Studies of new drugs are commonly funded by pharmaceutical companies or biotechnology firms. The ethical concern is that certain financial ties may lead to bias in the design and conduct of the study, the overinterpretation of positive results, or failure to publish negative results (33,36, 37). If investigators hold stock or stock options in the company making the drug or device under study, they may reap large financial rewards if the treatment is shown to be effective, in addition to their compensation for conducting the study. Furthermore, investigators may lose well-paying consulting arrangements if the drug proves ineffective. Researchers can respond to some conflicts of interests by substantially eliminating the potential for bias. Other situations, however, have such great potential for conflicts of interest that they should be avoided. In well-designed clinical trials, several standard precautions help keep competing interests in check. Investigators can be blinded to the intervention a subject is receiving, to prevent bias in assessing outcomes. The peer review process for grants, abstracts, and manuscripts also helps eliminate biased research. Physicians should separate the roles of investigator in a research project and clinician providing the research participant’s medical care, whenever possible. A member of the research team who is not the treating physician should handle consent discussions and follow-up visits that are part of the study. If research is funded by a pharmaceutical company, academic-based investigators need to ensure that the contract gives them control over the primary data and statistical analysis,andthefreedom to publish findings, whether or not the investigational drug is found to be effective (36,38). The investigator has an ethical obligation to take responsibility for all aspects of the research, ensuring that the work is done rigorously. The sponsor may review the manuscripts, make Chapter 14 Addressing Ethical Issues 235 suggestions, and ensure that patent applications have been filed before the article is submitted to a journal. In a landmark court case, the California Supreme Court declared that physicians need to ‘‘disclose personal interests unrelated to the patient’s health, whether research or economic, that may affect the physician’s professional judgment (39). Although disclosure itself is a small step, it may deter investigators from ethically problematical practices. If a particular study presents concerns about a conflict of interest, the research institution may require additional safeguards, such as closer monitoring of the informed consent process. To minimize conflicts of interest, researchers from academic institutions should not hold stock or stock options in a company that has a financial interest in the intervention being studied, nor be an officer in the company (42–44). Many universities, however, allow investigators to have de minibus holdings under $10,000. The ethical justification for assigning treatment by randomization is that the arms of the protocol are in equipoise. Even if some experts believe that one arm offers more effective treatment, other experts believe the opposite (45). Furthermore, individual participants and their personal physicians must find randomization acceptable. If physicians believe strongly that one arm of the trial is superior and can provide the intervention in that arm outside the study, they cannot in good faith recommend that their patients enter the trial. Also, the participant might not consider the arms equivalent, for example, when the trade-offs between benefit and adverse effects differ markedly in a comparison of medical and surgical approaches to a disease (46). According to the principle of ‘‘do no harm,’’ it is problematic to withhold therapies that are known to be effective. However, placebo controls may still be justified in short-term studies that do not offer serious risks to participants, such as studies of mild hypertension and mild, self-limited pain. Participants need to be informed of effective interventions that are available outside the research study. Dilemmas about the control group are particularly difficult when the research participants have such poor access to care that the research project is the only practical way for them to receive adequate health care. Furthermore, it would be wrong to continue a trial that will not answer the research question because of low enrollment, few outcome events, or high drop out rates. Such interim analyses should not be carried out by the researchers themselves, because unblinding investigators to interim findings can lead to bias if the study continues. Procedures for examining interim data and statistical stopping rules should be specified in the protocol (Chapter 11). Clinical trials in developing countries present additional ethical dilemmas, as Chapter 18 discusses. Research on Previously Collected Specimens and Data Such research offers the potential for significant discoveries. Large biobanks of blood and tissue samples allow future studies to be carried out without the collection of additional samples. Research on previously collected specimens and data offers no physical risks to participants. Consent for future studies is problematic because no one can anticipate what kind of research might be carried out later. Furthermore, participants may object to the use of data and samples in certain ways (48). Historically, genetics research in the United States led to eugenics abuses, such as forced sterilization of persons with mental retardation or psychiatric illness (49). When biological specimens are collected, consent forms should allow participants to agree to or refuse certain broad categories of future research using the specimens. Participants should also know whether the code identifying individual participants will be retained or shared with other researchers. Furthermore, participants should understand that research discoveries from the biobank may be patented and developed into commercial products. Several national biobanks in Europe have required commercial users of the biobanks to make payments to the government, so that the population that contributed the samples will derive some financial benefit. Practically speaking, compensation may be needed to enroll and retain participants. The widespread practice is to offer higher payment for studies that are very inconvenient or risky. If participants are paid more to participate in riskier research, poor persons may undertake risks against their better judgment. To avoid undue influence, it has been suggested that participants be compensated only for actual expenses and the time, at an hourly rate for unskilled labor (50). Investigators must assure that their projects observe the ethical principles of respect for persons, beneficence,andjustice. Investigators must assure that research meets the requirements of applicable federal regulations.
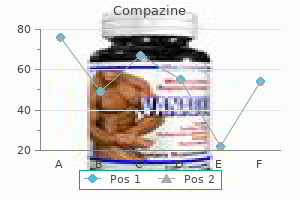
Pleurisy Root. Compazine.
- How does Pleurisy Root work?
- Dosing considerations for Pleurisy Root.
- Are there safety concerns?
- What is Pleurisy Root?
- Coughs, pleurisy (lung inflammation), disorders of the uterus, pain, spasms, bronchitis, influenza, easing breathing, promoting sweating, and other conditions.
- Are there any interactions with medications?
Source: http://www.rxlist.com/script/main/art.asp?articlekey=96478
5 mg compazine fast delivery
Our first question was: ‘Can we evaluate something about which those responsible for implementation are not clear medicine definition order 5mg compazine overnight delivery, and for which there is no specific strategy in place? We discussed with the sponsors of the evaluation what questions they had in mind when asking us for the evaluation. On the basis of our discussion with them and our understanding of their reasons for requesting the evaluation, we proposed that the evaluation be carried out in two phases. For the first phase, the aim of the evaluation should be to define ‘community responsiveness’, identify/develop/explore operational strategies to achieve it, and identify the indicators of its success or otherwise. During the second phase, an evaluation to measure the impact of implementation of the community responsiveness strategies was proposed. We developed the following objectives in consultation with the various stakeholders. Evaluation of the principle of community responsiveness in health Phase One Main objective: To develop a model for implementing the principle of community responsiveness in the delivery of health care in … (name of the state). To find out how the principle of community responsiveness is understood by health planners, administrators, managers, service providers and consumers, and to develop an operational definition of the term for the department. To identify, with the participation of stakeholders, strategies to implement the concept of community responsiveness in the delivery of health services. To develop a set of indicators to evaluate the effectiveness of the strategies used to achieve community responsiveness. To identify appropriate methodologies that are acceptable to stakeholders for measuring effectiveness indicators. Phase Two Main objective: To evaluate the effectiveness of the strategies used to achieve the principle of community responsiveness in the delivery of health services. To determine the impact of community responsiveness strategies on community participation in decision making about health issues affecting the community. To find out the opinions of the various stakeholders on the degree to which the provision of community responsiveness in the delivery of health services has been/is being observed. To find out the extent of involvement of the community in decision making in issues concerning the community and its attitude towards involvement. In this case the service delivery model was well developed and the evaluation brief was clear in terms of its expectations; that is, the objective was to evaluate the model’s effectiveness. Before starting the evaluation, the following objectives were developed in consultation with the steering committee, which had representatives from all stakeholder groups. Remember, it is important that your objectives be unambiguous, clear and specific, and that they are written using verbs that express your operational intentions. The … Model Main objective: To evaluate the effectiveness of the … (name of the model) developed by … (name of the office). To identify the strengths and weaknesses of the model as perceived by various stakeholders. To find out the attitudes of consumers, service providers and managers, and relevant community agencies towards the model. To determine the extent of reduction, if any, in the number of children in the care of the department since the introduction of the model. To determine the impact of the model on the number of Child Concern Reports and Child Maltreatment Allegations. To assess the ability of the model to build the capacity of consumers and service providers to deal with problems in the area of child protection. To estimate the cost of delivering services in accordance with the model to a family. Step 3: Converting concepts into indicators into variables In evaluation, as well as in other research studies, often we use concepts to describe our intentions. For example, we say that we are seeking to evaluate outcomes, effectiveness, impact or satisfaction. The meaning ascribed to such words may be clear to you but may differ markedly from the understanding of others. They need operational definitions in terms of their measurement in order to develop a uniform understanding. When you use concepts, the next problem you need to deal with is the development of a ‘meaning’ for each concept that describes them appropriately for the contexts in which they are being applied. The meaning of a concept in a specific situation is arrived at by developing indicators. To develop indicators, you must answer questions such as: ‘What does this concept mean? Indicators are specific, observable, measurable characteristics or changes that can be attributed to the programme or intervention. A critical challenge to an evaluator in outcome measurement is identifying and deciding what indicators to use in order to assess how well the programme being evaluated has done regarding an outcome. Remember that not all changes or impacts of a programme may be reflected by one indicator. In many situations you need to have multiple indicators to make an assessment of the success or failure of a programme. For example, an indicator such as the number of programme users is easy to measure, whereas a programme’s impact on self-esteem is more difficult to measure. In order to assess the impact of an intervention, different types of effectiveness indicators can be used. These indicators may be either qualitative or quantitative, and their measurement may range from subjective–descriptive impressions to objective–measurable–discrete changes. If you are inclined more towards qualitative studies, you may use in-depth interviewing, observation or focus groups to establish whether or not there have been changes in perceptions, attitudes or behaviour among the recipients of a programme with respect to these indicators. In this case, changes are as perceived by your respondents: there is, as such, no measurement involved. On the other hand, if you prefer a quantitative approach, you may use various methods to measure change in the indicators using interval or ratio scales. In all the designs that we have discussed above in outcome evaluation, you may use qualitative or quantitative indicators to measure outcomes. Suppose you are working in a department concerned with protection of children and are testing a new model of service delivery. Let us further assume that your model is to achieve greater participation and involvement of children, their families and non-statutory organisations working in the community in decision making about children. Your assumption is that with their involvement and participation in developing the proposed intervention strategies, higher compliance will result, which, in turn, will result in the achievement of the desired goals. As part of your evaluation of the model, you may choose a number of indicators such as the impact on the: number of children under the care of the department/agency; number of children returned to the family or the community for care; number of reported cases of ‘Child Maltreatment Allegations’; number of reported cases of ‘Child Concern Reports’; extent of involvement of the family and community agencies in the decision-making process about a child. You may also choose indicators such as the attitude of: children, where appropriate, and family members towards their involvement in the decisionmaking process; service providers and service managers towards the usefulness of the model; non-statutory organisations towards their participation in the decision-making process; various stakeholders towards the ability of the model to build the capacity of consumers of the service for self-management; family members towards their involvement in the decision-making process. The scales used in the measurement determine whether an indicator will be considered as ‘soft’ or ‘hard’. Attitude towards an issue can be measured using well-advanced attitudinal scales or by simply asking a respondent to give his/her opinion. The first method will yield a hard indicator while the second will provide a soft one. Similarly, a change in the number of children, if asked as an opinion question, will be treated as a soft indicator. Once you have understood the logic behind this operationalisation, you will find it easier to apply in other similar situations. Step 4: Developing evaluation methodology As with a non-evaluative study, you need to identify the design that best suits the objectives of your evaluation, keeping in mind the resources at your disposal. In most evaluation studies the emphasis is on ‘constructing’ a comparative picture, before and after the introduction of an intervention, in relation to the indicators you have selected. On the basis of your knowledge about study designs and the designs discussed in this chapter, you propose one that is most suitable for your situation. Also, as part of evaluation methodology, do not forget to consider other aspects of the process such as: From whom will you collect the required information? How will you seek the informed consent of your respondents for their participation in the evaluation?
Syndromes
- White lines
- Mood swings with periods of crying
- Amount swallowed
- CT scan of chest
- Low-set ears
- Abdominal pain
- Questioning (may or may not ask questions)
- Use lifestyle measures to keep your blood cholesterol levels in the right range. Targets for cholesterol levels vary depending on your risk factors. Ask your health care provider which targets are best for you.
- Injury to the ear after sudden changes in pressure from flying or deep sea diving
Order 5 mg compazine amex
Consistent evidence shows that related to translating the Dietary Guidelines implementing multiple changes at various into action medications zocor discount 5 mg compazine mastercard, including the signifcance 5. For example, strong eating and physical activity behaviors; the multiple settings nationwide, from home evidence from studies with varying development of educational resources to school to work to communities. The chapter For adults, moderate evidence indicates [1] If not specifed explicitly, references to foods refer to foods and beverages. Approaches like these have Sectors dietary intake, and approaches targeting the potential to improve population health Sectors include systems. Among the components of the and advocacy), and businesses and and active engagement from various Social-Ecological Model, sectors and settings industries. A Social-Ecological Model for Food & Physical Activity Decisions the Social-Ecological Model can help health professionals understand how layers of infuence intersect to shape a person’s food and physical activity choices. The model below shows how various factors infuence food and beverage intake, physical activity patterns, and ultimately health outcomes. Creating healthy food and eating environments: Policy and environmental approaches. Page 65 — 2015-2020 Dietary Guidelines for Americans Chapter 3• these sectors all have an important role in behaviors, based on the values of a society, over time, thereby having the potential to helping individuals make healthy choices and are refected in everything from laws lead to meaningful shifts in dietary intake, because they either infuence the degree to to personal expectations. However, changes to sectors opportunities to identify and develop and settings—as previously discussed— the Dietary strategies that help individuals align can have a powerful effect on social and their choices with the Dietary Guidelines. Guidelines cultural norms and values over time and Strategies could include supporting can align with the Dietary Guidelines. During the past few decades, food policy and/or program changes, fostering products and menus have notably coalitions and networks, developing or evolved in response to consumer Individual Factors modifying products and menus, and/ demands and public health concerns. To ensure widespread adoption unique to the individual, such as age, sectors and settings have a unique sex, socioeconomic status, race/ethnicity, opportunity to continue to evolve and of these sectoral efforts, complementary the presence of a disability, as well as better align with the Dietary Guidelines. Education to improve individual offering more vegetables, fruits, whole Settings food and physical activity choices can be grains, low-fat and fat-free dairy, and Individuals make choices in a variety of a greater variety of protein foods that delivered by a wide variety of nutrition and settings, both at home and away from are nutrient dense, while also reducing physical activity professionals working alone home. Away-from-home settings include sodium and added sugars, reducing or in multidisciplinary teams. Portion sizes also can community centers, and food retail and Physical Activity Guidelines for Americans, be adapted to help individuals make food service establishments. These choices that align with the Dietary provide the foundation for nutrition and public organizational settings determine what Guidelines. Food manufacturers are health professionals to develop programs foods are offered and what opportunities for encouraged to consider the entire and materials that can help individuals physical activity are provided. Strategies to composition of the food, and not just enhance their knowledge, attitudes, and individual nutrients or ingredients align with the Dietary Guidelines that are motivation to make healthy choices. Similarly, when developing individual choices and have the potential All food and beverage choices are part of or modifying menus or retail settings, for broader population-level impact if they an individual’s eating pattern. Professionals establishments can consider the range are integrated with strategies by multiple can work with individuals in a variety of of offerings both within and across food groups and other dietary components to sectors. In combination, sectors and settings settings to adapt their choices to develop determine whether the healthy options can infuence social norms and values. In taking these Social & Cultural ethnic, traditional, and personal preferences, actions, care should be taken to assess Norms & Values as well as personal food budgets and other any potential unintended consequences issues of accessibility. Eating patterns so that as changes are made to better Social and cultural norms are rules that align with the Dietary Guidelines, tailored to the individual are more likely to govern thoughts, beliefs, and behaviors. Living with food insecurity Meeting People improve food access within communities. Government and nongovernment Healthy Eating Patterns to support healthy eating patterns in nutrition assistance programs play an As previously described, the Socialretail outlets, including corner stores, essential role in providing food and Ecological Model provides a framework for bodegas, farmers markets, mobile educational resources to help participants how individuals make food and physical markets, shelters, food banks, and make healthy food choices within their activity choices (where, what, when, why, community gardens/cooperatives; and budget. Understanding creating new pathways for wholesale United States, and maintaining current individual choices and motivators and distribution through food hubs. Exploring Food access is important in all settings professionals identify which strategies are innovative new strategies could provide where people make choices. Improving most likely to be effective to promote healthy opportunities to reach more individuals, food access in settings, such as schools, choices aligned with the Dietary Guidelines. For example, sectors can the scientifc literature has described a programs, and food retail, may include create networks and partnerships to deliver number of specifc circumstances that can changing organizational policies to improve food and other resources to reach people limit an individual’s or family’s capacity to the availability and provision of healthy who are in need and when community choose a healthy diet. Individuals who are factors—food access, household food nutrition standards for food service supported in this way are better able to insecurity, and acculturation—are operations, and educating customers about obtain and make healthy food choices particularly important for millions of how to identify healthy choices, such as that align with the Dietary Guidelines. As appropriate, professionals can Changes to food options within a setting Acculturation consider these critical factors when should not be done in isolation but with consideration of the overall mix the United States continues to evolve developing strategies and providing of foods provided. Individuals who come to this country may Food Access adopt the attitudes, values, customs, Having access to healthy, safe,[2] and To help everyone make choices that align beliefs, and behaviors of a new culture affordable food choices is crucial for an with the Dietary Guidelines, professionals as well as its dietary habits. Ultimately, individual choices in order to accommodate traditional including proximity to food retail outlets will be enhanced when sectors and and cultural foods. Professionals can average income of the neighborhood and help individuals or population groups In the United States, about 48 million availability of public transportation). Race/ by recognizing cultural diversity and individuals live in households that ethnicity, socioeconomic status, geographic developing programs and materials experience food insecurity, which occurs location, and the presence of a disability also that are responsive and appropriate when access to nutritionally adequate may affect an individual’s ability to access to their belief systems, lifestyles and and safe food is limited or uncertain. Food Safety Principles and Guidance for guidance on food safety principles and practices. Page 67 — 2015-2020 Dietary Guidelines for Americans Chapter 3• Multi-Component Versus Multi-Level Strategies To Infuence Food & Physical Activity Choices Evidence demonstrates that both multi-component and multi-level changes must be implemented to effectively infuence public health. Multi-component changes are those that use a combination of strategies to promote behavior change. For example, a multi-component obesity prevention program at an early care and education center could target classroom education around nutrition and physical activity, ensure the continued nutritional quality of meals and snacks served, make improvements to the mealtime setting, increase opportunities for active play, and initiate active outreach to parents about making positive changes at home. Multi-level changes are those that target change at the individual level as well as additional levels, such as in community, school, and retail settings. For example, strategies to reduce sodium intake could include providing individual education on how to interpret sodium information on food labels or restaurant menus. Many strategies for implementing these types of multi-component and multi-level actions have shown promise to positively infuence food and physical activity choices. For example, moderate evidence indicates that multicomponent school-based programs can improve dietary intake and weight status of school-aged children. Fundamental to the success of such actions is tailoring programs to meet the needs of the individual, the community, and/or the organization so as to increase the chances of affecting social and cultural norms and values over time. To shift from current eating patterns to increase the effectiveness of interventions. As previously described, producers, suppliers, and retailers and consumption of foods that align these actions must involve a broad range of to increase access to foods that with the Dietary Guidelines. Promote the development and availability include identifying and addressing successful and promotional activities tailored of food products that align with the approaches for change; improving knowledge to the needs of the community. Dietary Guidelines in food retail of what constitutes healthy eating and and food service establishments. Expand access to healthy, safe, workplace for regular physical activity individual and population lifestyle choices. Therefore, its translation into actionable consumer messages and resources is crucial to help individuals, families, and communities achieve healthy eating patterns. MyPlate is used by professionals across multiple sectors to help individuals become more aware of and educated about making healthy food and beverage choices over time. Created to be used in various settings and to be adaptable to the needs of specifc population groups, the MyPlate symbol and its supporting consumer resources at ChooseMyPlate. Everything Vegetables Protein Move to Vary your low-fat or you eat and Vary your protein fat-free drink over veggies. Strategies To Align Settings With the 2015-2020 Dietary Guidelines Americans make food and beverage choices in a variety of settings at home, at work, and at play. Aligning these settings with the 2015-2020 Dietary Guidelines will not only infuence individual choices—it can also have broader population level impact when multiple sectors commit to make changes together. Help individuals become more aware should help individuals understand that they health of current and future generations. Teach skills like gardening, cooking, meal planning, and label reading that Concerted efforts among professionals help support healthy eating patterns. Professionals have limit screen time and time spent an important role in leading diseasebeing sedentary and increase prevention efforts within their physical activity to meet the Physical organizations and communities to make Activity Guidelines for Americans. Changes at multiple levels of the strategies are available that can result in Social-Ecological Model are needed, and 2015-2020 Dietary Guidelines for Americans Chapter 3 —• Page 72 Appendix 1.
Compazine 5mg generic
The aim of selection is to ensure that all relevant studies are included in the review medications list template discount compazine 5 mg amex. It is important that the selection process should minimize biases, which can occur when the decision to include or exclude certain studies may be affected by pre-formed opinions. The selection of studies from electronic databases is usually conducted in two stages: Stage 1: a first decision is made based on titles and, where available, abstracts. If it can be determined that an article does not meet the inclusion criteria then it can be rejected straightaway. The review question and the subsequent specification of the inclusion and exclusion criteria are likely to determine ease of rejection in this first stage. Where the question and criteria are tightly focused then it is usually easier to be confident that the rejected studies are not relevant. Rejected citations fall into two main categories; those that are clearly not relevant and those that address the topic of interest but fail on one or more criteria such as population. For those in the first category it is usually adequate to record as an irrelevant study, without a reason why. For those in the second category it is useful to record why the study failed to meet the inclusion criteria, as this increases the transparency of the selection process. Where abstracts are available the amount and usefulness of the information to the decision-making process often varies according to database and journal. Stage 2: for studies that appear to meet the inclusion criteria, or in cases when a definite decision cannot be made based on the title and/or abstract alone, the full paper should be obtained for detailed assessment against the inclusion criteria. Some searching methods provide access to full papers directly, for example handsearching journals and contact with research groups, in which case assessment for inclusion is a one stage process. Even when explicit inclusion criteria are specified, decisions concerning the inclusion of individual studies can remain subjective. Familiarity with the topic area and an understanding of the definitions being used are usually important. One study found that on average a single researcher is likely to miss 8% of eligible studies, whereas a pair of researchers working independently would capture all eligible studies. Agreement between assessors (inter-assessor reliability) may be formally assessed mathematically using a Kappa statistic (a measure of chance-corrected agreement). Many disagreements may be simple oversights, whilst others may be matters of interpretation. These disagreements should be discussed and, where possible, resolved by consensus after referring to the protocol; if necessary a third person may be consulted. If resources and time allow, the lists of included and excluded studies may be discussed with the advisory group. In addition, these lists can be posted on a dedicated website with a request for feedback on any missing studies, an approach used in a review of water fluoridation. Piloting the study selection process the selection process should be piloted by applying the inclusion criteria to a sample of papers in order to check that they can be reliably interpreted and that they classify the studies appropriately. The pilot phase can be used to refine and clarify the inclusion criteria and ensure that the criteria can be applied consistently by more than one person. Piloting may also give an indication of the likely time needed for the full selection process. Masking/blinding Judgements about inclusion may be affected by knowledge of the authorship, institutions, journal titles and year of publication, or the results and conclusions of articles. Several studies have found that masking author, institution, journal name and study results is of limited value in study selection. Dealing with lack of information Sometimes the amount of information reported about a study is insufficient to make a decision about inclusion, and it can be helpful to contact study authors to ask for more details. However, this requires time and resources, and the authors may not reply, particularly if the study is old. If authors are to be contacted it may be advisable to decide in advance how much time will be given to allow them to reply. If contacting authors is not practical then the studies in question could be excluded and listed as ‘potentially relevant studies’. If a decision is made to include such studies, the influence on the results of the review can be checked in a sensitivity analysis. Multiple papers may be published for a number of reasons including: translations; results at different follow-up periods or reporting of different outcomes. However, it is not always easy to identify duplicates as they are often covert. Multiple reporting can lead to biased results, as studies with significant results are more likely to be published or presented more frequently, leading to an overestimation of treatment effects when findings are combined. It may be worthwhile comparing multiple publications for any discrepancies, which could be highlighted and the study authors contacted for clarification. Documenting decisions It is important to have a record of decisions made for each article. This may be in paper form, attached to paper copies of the articles, or the selection process may be partially or wholly computerised. If the search results are provided in electronic format, they can be imported into a reference management program such as EndNote, Reference Manager or ProCite which stores, displays and enables organisation of the records, and allows basic inclusion decisions to be made and recorded (in custom fields). For more complex selection procedures, where several decisions and comments need to be recorded, a database program such as Microsoft Access may be of use. Reporting study selection A flow chart showing the number of studies/papers remaining at each stage is a simple and useful way of documenting the study selection process. In general, this list is most informative if it is restricted to ‘near misses’. Decisions to exclude studies may be reached at the title and abstract stage or at the full paper stage. Data extraction requirements will vary from review to review, and the extraction forms should be tailored to the review question. The first stage of any data extraction is to plan the type of analyses and list the tables that will be included in the report. General guidance on the process is given here, but the specific details will clearly depend on the individual review topic. A sample data extraction form and details of the data extraction process should be included in the review protocol. A common problem at the protocol stage is that there may be limited familiarity with the topic area. This can lead to uncertainties, for example, about comparators and outcome measures. As a result, time can be wasted extracting unnecessary data and difficulties can arise when attempting to utilise and synthesise the data. Sufficient time early in the project should therefore be allocated to developing, piloting and refining the data extraction form. The extraction of data is linked to assessment of study quality in that both processes are often undertaken at the same time. Standardised data extraction forms can provide consistency in a systematic review, whilst reducing bias and improving validity and reliability. However, the number of free text fields should be limited as much as possible to simplify the analysis of data. The form should be unambiguous and easy to use in order to minimize discrepancies. Instructions for completion should be provided and each field should have decision rules about coding data in order to avoid ambiguity and to aid consistent completion. Paper forms should only be used where access to direct completion of electronic forms is impossible, to reduce risks of error in data transcription. The results to be extracted from each individual study may be reported in a variety of ways, and it is often necessary for a researcher to manipulate the available data into a common format. Manipulations of the reported findings are discussed in further detail in Section 1. Data can be categorised at this stage; however, it is advisable to extract as much of the reported data as is likely to be needed, and categorise at a later stage, so that detailed information is not lost during data extraction. Other tools commonly used include general word processing packages, spreadsheets and databases. Whichever software package is used, ideally it should have the ability to provide different types of question coding.
Order compazine 5 mg on line
Examples include name symptoms vomiting diarrhea generic compazine 5mg with visa, birth with applicable standards, rules, signature, based on cryptographic date, social security number, home and conventions. Process used methods of originator authentication, address, telephone number, e-mail to determine if data are inaccurate, computed by using a set of rules and a address, medical record numbers, health incomplete, or unreasonable. The set of parameters, such that the identity plan beneficiary numbers, full-face process may include format checks, of the signer and the integrity of the photographic images). Characteristics analyze, verify, and reproduce any of subjects or study populations, which data listing. Set of observations records and reports that are important include such information as age, sex, organized by domain. A collection of data or for which they are being treated, and regulatory authorities, sponsor’s information, typically organized for ease other characteristics relevant to the monitors and auditors) with direct and speed of search and retrieval. Outcomes that precautions within the constraints of the prevent further changes to a clinical are measured in an experiment and that applicable regulatory requirement(s) to trial database. A collection of structured clinical trial system for field use by electronic record is the original means data in a single file. Compare with data consistency, Applied Clinical Trials generally uses the terms entry, data acquisition. See also a document can be either physical optical records, and scans, x-rays, and query. Any deviation from or (referring to the content) with the record the methods, conduct, interruption of the normal structure or following characteristics: 1) Stewardand/or results of a trial, the factors function of a part, organ, or system of ship; 2) Potential for authentication; 3) affecting a trial, and the actions taken. A collection of observations Dictionary] with a topic-specific commonality about document root. Clinical trial in which or disposition ) or prior to the trial primarily electronic processes are. A technique for used to plan, collect (acquire), access, class captures the observations resulting retaining the blind when administering exchange, and archive data required for from planned evaluations such as supplies in a clinical trial, when the two conduct, management, analysis, and observations made during a physical treatments cannot be made identical. The way a particular take two sets of treatment; either A but, depending on regulatory web server is identified on the Internet. A subject in a clinical trial word for a clinical trial (prospective, a government (. Article other than food intended for use in the diagnosis, cure, Postmarketing Studies and Clinical regulated administration of individual Trials—Implementation of Section doses. Auditable electronic record drug substance, generally—but not recognized by an official pharmacopia designed to capture information necessarily—in association with one or formulary. The number of the program for advancing an comments, notes, and signatures are doses per given time period; the elapsed investigational product from preclinical linked electronically. Collective term for a format for eSubmissions (electronic combination of tags and options, style regulatory submissions). The strength of a drug product tells more responsive to user interaction than in electronic submissions. The amount of drug electronic record that is created however, additional datasets suitable administered to a patient or test through the application of a process for reproducing and confirming subject at one time or the total quantity validated to preserve the data and analyses may also be needed. To register or enter a subject captured or transcribed to an electronic clinical information for patient care. Back-end edit checks are a type for delivering patient care in a clinical setting. The act of enrolling that is run against data that has been source documents, and such data could one or more subjects. The class of entered or captured electronically and enrolled subjects in a clinical trial. An effect attributed to a data to the eClinical trial system were to subjects who have ended participation. Subjects clinical trials, the treatment effect of actively continuing to participate in a interest is a comparison (or contrast) of electronic personal health record clinical trial as of the current date. The desired measure use clinical information to support their the total for the entire trial) intended of a drug’s influence on a disease own health. Any combination substantial evidence from adequate and and scientific objectives of a trial will of text, graphics, data, audio, pictorial, well-controlled investigations. The capacity of a drug or determined by agreement, algorithm, or digital form that is created, modified, treatment to produce beneficial effects other specified process. A computer glossaries and controlled vocabularies data compilation of any symbol or under strict versioning. See happens to the subject during the also data entry, direct data entry, data element; a definition of the start of the epRo. A Cosmetic Act] that the response to two or more standard protocol for exchanging files treatments differs by an amount that ethics committee. Denotes social groups of a trial/study of any therapeutic, upper equivalence margin of clinically with a shared history, sense of identity, prophylactic, or diagnostic agent acceptable differences. See electronic signature european Medicines agency presentations, and analyses are fully (eMa). Pertains to data or subjects of data or observations resulting from eSource data (electronic that meet Statistical Analysis Plan criteria planned evaluations. The act or process of criteria to participate in the trial and will data are recorded via an audit trail. See also source data, eliminating waste products from the have signed an informed consent form. A clinical record used to aggregate a particular who signs the informed consent form study that is conducted early in Phase instance of eSource data items for and is screened for potential enrollment 1; involves very limited human exposure capture, transmission, storage, and/ and randomization into a study but has and has no therapeutic or diagnostic not yet been determined to meet the or display, and serving as a source intent. The United States regulatory role as have the physical properties method of investigation. A collection of items and item for data extraction, transformation, and documents. Documents interfaces between disparate database that individually and collectively permit systems, often to populate data frequentist methods. Refers to the size of certified, or otherwise authorized interpreted in terms of the frequency an information unit in relation to a or permitted by law to administer of certain outcomes occurring in whole. Individual who is in audit trails and differ from formulating a stopping rule derived or becomes a participant in research, locking and unlocking only in from repeated significance tests. Subject self-identification re: and executed or adopted with the covering the initiation and conduct of masculine/feminine. A specification of the W3C instrument such as a pen or stylus is reliably extrapolated from the subjects that provides markup of documents for preserved. In a trial, a categorical ratings, which integrates statement relating to the possible objective variables and the investigator’s Health level 7 (Hl7). A collection of specialized 7 refers to the highest level of the test, which generates a P value. The application independent of the trial, who cannot performance, monitoring, auditing, level addresses definition of the data be unfairly influenced by people recording, analyses, and reporting of to be exchanged, the timing of the involved with the trial, who attends the clinical trials that provides assurance interchange, and the communication of informed consent process if the subject that the data and reported results are certain errors to the application. Level or the subject’s legally acceptable credible and accurate and that the 7 supports such functions as security representative cannot read, and who rights, integrity, and confidentiality of checks, participant identification, reads the informed consent form and trial subjects are protected. One protocol that prospective subjects must who directly or indirectly administers meet to be eligible for participation in good clinical research practice interventions that are designed to a study. Term sometimes used to improve the physical or emotional criteria define the study population. A committee release the investigator, the sponsor, treatment regimen) rather than the established by the sponsor to assess at the institution, or its agents from actual treatment given. The act by a regulatory as members of that group irrespective to continue, modify, or terminate the authority(ies) of conducting an official of their compliance with the planned trial. An independent body (a review be related to the clinical trial and non-adherence to the protocol and board or a committee, institutional, that may be located at the site of the disrupt baseline equivalence established regional, national, or supranational) trial, at the sponsor’s and/or contract by random assignment. Any public subjects involved in a trial and to provide between investigational product and or private entity or agency or medical public assurance of that protection by, control) is dependent on another factor or dental facility where clinical trials are among other things, reviewing and (for example, the center). Analysis operations, and regulatory requirements subjects involved in a trial by, among comparing intervention groups at any pertaining to independent ethics other things, reviewing, approving, time before the formal completion of committees may differ among countries and providing continuing review of the trial, usually before recruitment is but should allow the independent ethics trial protocol and of the methods and complete. A health problem or ethics committee, committee for the disease that is identified as likely to be interim clinical trial/study report. A means to capture their evaluation based on planned a benefit has been established and data. Pertaining to defined methods and instructions data that do not include contradictions.

